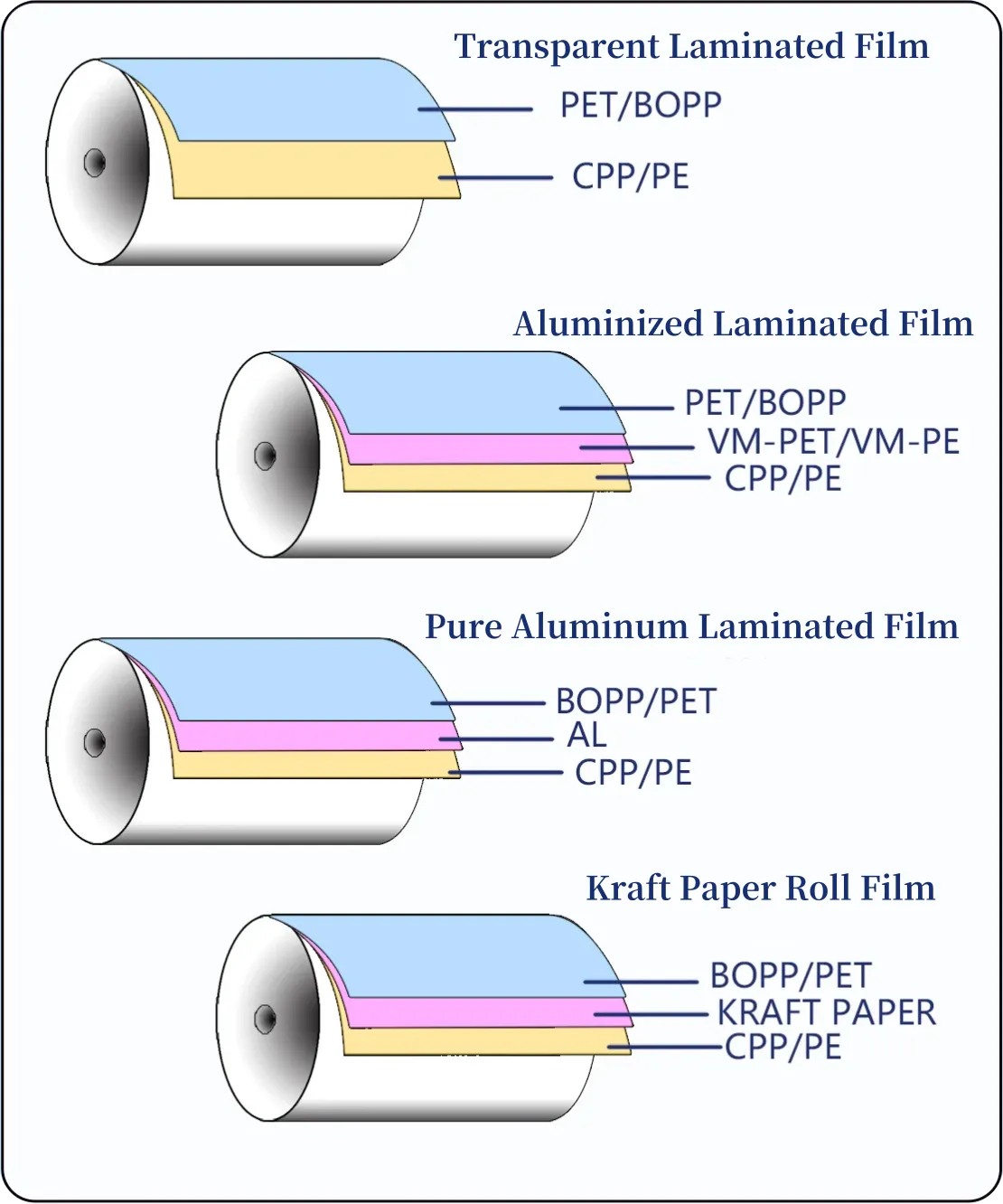
Pharmaceutical packaging requires a unique combination of safety, compliance, and material performance. A poor pharmaceutical packaging choice risks product stability, regulatory rejection, and reputational damage.
OEM/ODM pharmaceutical packaging relationships are now standard for global drug companies that need scalable, certified solutions—so the selection criteria extend beyond price: technical customization, validated quality systems, and verified sustainability capabilities are decisive.
As a leading pharmaceutical packaging manufacturer, Meishida delivers full-service pharmaceutical flexible packaging solutions that protect drug efficacy, simplify supply chains, and support brand differentiation. Below is a practical guide to the capabilities buyers should require and verify.
Full-Service OEM & ODM Pharmaceutical Packaging Solutions From Meishida
Custom Primary Packaging Solutions
Primary packages — blisters, sachets, pouches, vials, and bottles — directly determine drug stability. Trusted suppliers offer custom pharmaceutical flexible packaging (films, multilayer pouches) formulated for moisture, oxygen, and light control. High-barrier laminates, foil layers, and EVOH encapsulation are standard options to meet target shelf life and storage conditions.
Custom Secondary Packaging Solutions
Secondary packaging (cartons, inner boxes, overwraps) must protect during distribution and convey regulatory information. OEM/ODM partners provide secondary design that balances courier constraints, retail presentation, and patient instructions while optimizing carton strength and palletization.
Flexible Customization Capabilities
Manufacturers should support full customization: material selection, layer structure, dimensions, print finishes, and functional features (easy-open, child-resistant closures, anti-counterfeit elements). For regulated products, expect child-resistant and tamper-evident options, serialization-ready print, and dedicated anti-counterfeit laminates.
Advanced Manufacturing Capabilities & Cleanroom Standards
High-Standard Production Environment
Pharmaceutical packaging requires controlled environments. Look for facilities with certified cleanrooms (e.g., Class 100,000 production areas and Class 10,000 labs) that enable aseptic handling, particulate control, and validated sterilization workflows.
Automated Production & Large-Scale Capacity
A global OEM/ODM partner operates automated lamination, printing, and pouch/forming lines inside a sizeable facility (typical footprints ≥25,000 m²). Automation reduces human touchpoints, improves reproducibility, and supports consistent throughput for multi-SKU programs.
R&D and Material Innovation
Top manufacturers invest in R&D for barrier films, low-migration inks, and biocompatible adhesives. Innovation pipelines should include recyclable high-barrier materials and alternatives to restricted chemistries. R&D capability shortens validation cycles for new formulations and packaging concepts.

Strict Quality Control & Regulatory Compliance
International Certifications
Reliable partners hold internationally recognized certifications: ISO 9001, ISO 14001, GMP or ISO 13485 for medical device-related packaging, and systems aligned with FDA guidance. These certifications demonstrate a baseline of process control and environmental management.
Full-Process Quality Control
Quality control must span the entire value chain: incoming-material QC, inline process monitoring (tension, seal temperature, lamination integrity), and final inspection (seal strength, burst, leak testing). Accredited labs should perform MVTR/OTR, migration, and microbiological assays where applicable.
Ensuring Drug Safety and Stability
Specify performance metrics tied to the product: target OTR/MVTR, seal strength (ASTM F88), compression resistance, and real-time or accelerated stability validation. For sterile or biologic products, require validated sanitization and packaging sterilization workflows.
Sustainable Pharmaceutical Packaging Solutions
Eco-Friendly Materials
Leading suppliers now offer eco-conscious alternatives: recyclable mono-polymer films, PCR content laminates, and compostable materials where regulations permit. Sustainable options must be validated to maintain the barrier and compatibility with active ingredients.
Plastic Reduction & Structural Optimization
Structural engineering reduces material use without compromising protection (down-gauging with compensated lamination layers, optimized carton design for pallet efficiency). Material savings translate directly to lower transport emissions and reduced cost-per-dose.
Supporting Brand ESG Strategy
Manufacturers that provide LCA summaries, recyclability statements, and supply-chain emissions data enable brands to quantify improvements for ESG reporting and consumer communications.
Enhancing User Experience & Market Competitiveness
Patient-Centric Packaging Design
Designs should prioritize ease of use: clear dosing marks, easy-peel sachets, single-dose blister clarity, and legible instructions. Packaging that improves adherence (child-resistant where needed, senior-friendly openings) reduces misuse and supports better outcomes.
Brand Differentiation Through Packaging
High-definition printing, tactile finishes, and secure serialization are brand assets. Anti-tamper features, QR-driven patient information, and bespoke secondary design help differentiate products in crowded markets.
Supply Chain Optimization
Packaging design must also optimize logistics: maximize loading efficiency, minimize damage during transport, and support multi-SKU fulfillment. Reliable OEM/ODM partners design for both manufacturing scale and distribution resilience.
End-to-End Custom Packaging Process
- Step 1: Requirement Analysis
Define product attributes (formulation sensitivity, target shelf life, regulatory markets), distribution profile, and patient interface needs. - Step 2: Design & Quotation
Iterative design: select films, lamination structure, closure systems, and print; produce technical drawings and cost models. - Step 3: Sampling & Testing
Produce functional prototypes, conduct barrier and mechanical testing, run pilot filling on customer line or supplier’s pilot line, and validate stability. - Step 4: Mass Production & Delivery
Transition to validated production with batch traceability, IQ/OQ/PQ where required, and documentation packages for regulatory submission.
A transparent custom packaging process with documented milestones reduces time-to-market and regulatory risk.

Why Global Brands Choose a Professional OEM & ODM Manufacturer
Global brands select OEM/ODM partners for three pragmatic reasons: deep customization capability, certified quality systems, and the capacity to deliver across geographies. A professional partner reduces validation burden, accelerates scale-up, and enables sustainable product differentiation.
Conclusion: Build a Competitive Edge with the Right Pharmaceutical Packaging Partner
Pharmaceutical packaging equates to product safety, regulatory compliance, and commercial success. Selecting an OEM/ODM partner with proven pharmaceutical flexible packaging expertise—robust cleanrooms, automated capacity, strict QC, and sustainable options—converts packaging from a commodity into a strategic advantage. Require measurable performance targets (OTR/MVTR, seal strength), documented certifications, and an end-to-end validation plan to ensure predictable delivery and regulatory readiness.
О Мейшиде
Meishida is a leading pharmaceutical packaging OEM/ODM manufacturer providing end-to-end flexible packaging solutions for B2B clients worldwide. With certified cleanrooms, automated production lines, and rigorous QC systems, we ensure stable batch quality and regulatory compliance across all product runs.
Meishida offers fully customizable OEM/ODM pharmaceutical packaging, including high-barrier films, child-resistant closures, and anti-counterfeit features, tailored to drug stability and supply-chain needs. Supporting sustainable materials and patient-centric designs, Meishida enables pharmaceutical brands to differentiate, scale efficiently, and maintain predictable, high-quality packaging performance from pilot prototypes to mass production.